HPLC Systems
Enhancing productivity by leveraging technologies

As workloads increase and businesses look to expand, analytical laboratories across multiple sectors are turning to the latest HPLC technologies to drive improvements in productivity.
The COVID-19 pandemic has upended many lives and businesses on an unprecedented scale. The analytical instrumentation sector is facing challenges in its manufacturing and supply chain, such as delivering products to end-users in a timely manner as well, as attending to an uneven demand for products and services.
The chromatography instruments market is facing a period of short-term negative growth, which can be attributed to factors like decline in the product demand from major end-users, limited operations in most of the industries, inadequate funding to research and academic institutes, temporary closure of major academic institutes, and disrupted supply chain and challenges in terms of providing essential/post-sales services.
However, the pandemic is not estimated to have long-term effects on the fundamental growth drivers in the chromatography products market. With many pharmaceutical and biopharmaceutical companies focusing on new drug development to combat the pandemic, the growth of the chromatography instruments market is likely to augment considerably in coming years.
Indian market. The market growth over the last couple of years has declined from 12 percent per annum to 5-6 percent per annum. This may be largely attributed to the FDA approvals that are pending for most vendors.
COVID-19 did not have much impact on the HPLC market. As expected, the AMJ 2020 quarter was a complete write-off, as far as instruments are concerned. Subsequently sales in OND 2020 saw a partial recovery. The pharma manufacturers restricted to buying equipment for COVID-related activities.
The high-end HPLC segment is dominated by Waters. Other players include Shimadzu, Thermo Fisher, Agilent, and PerkinElmer. These systems find applications primarily with mass spectrometry (MS) and front-end separations. Leading pharma companies as Pfizer and Glaxo also buy this product, their investment did not show much decline in 2020.
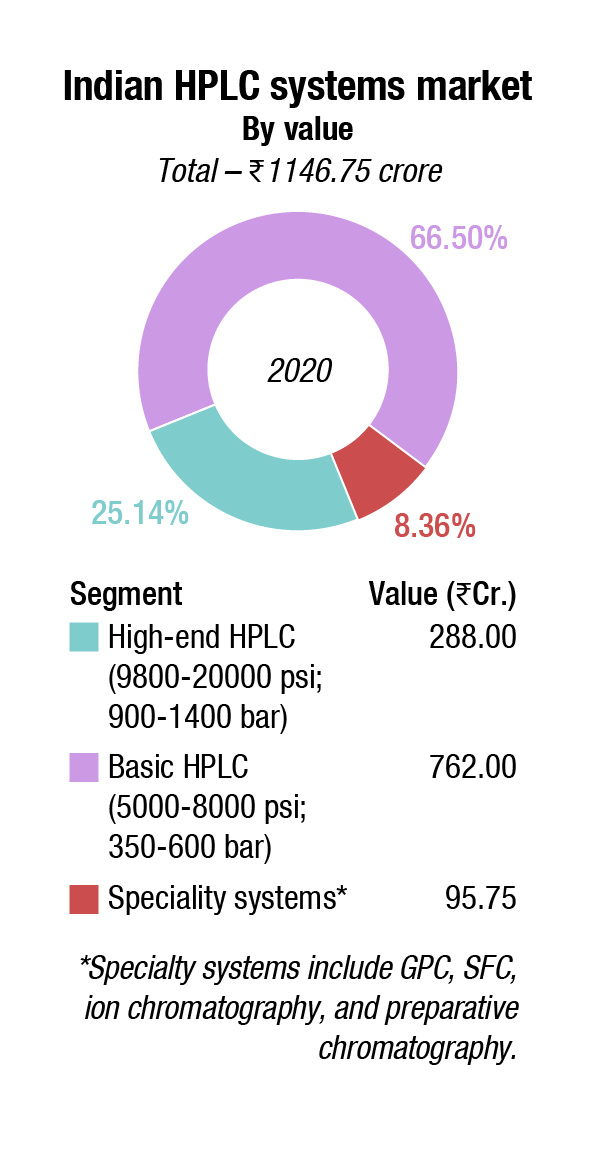
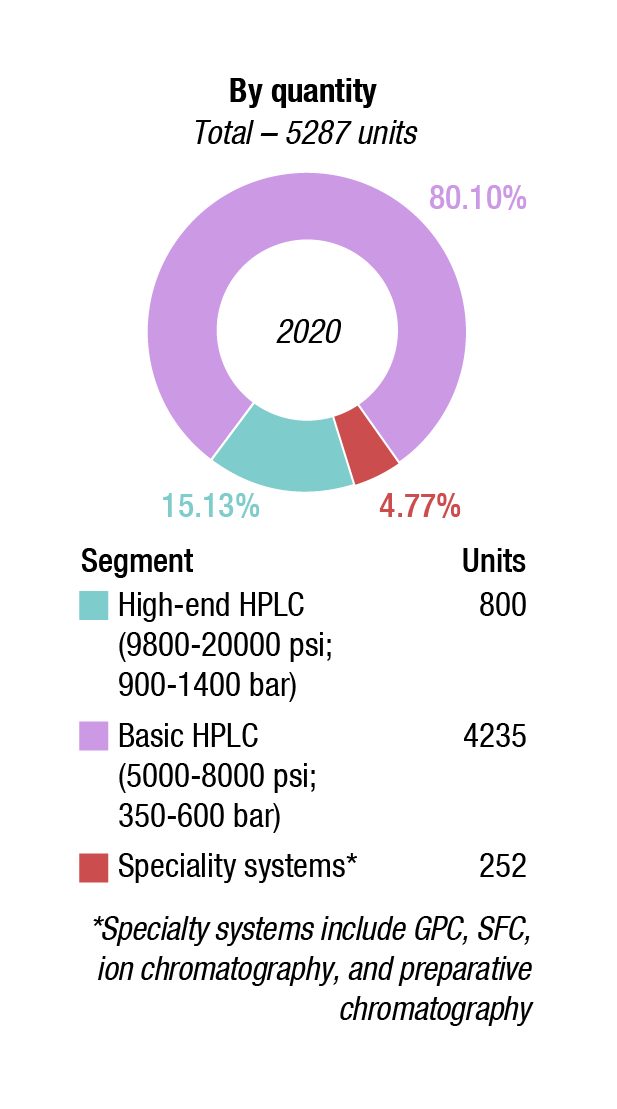
The basic HPLC systems saw a 5 percent decline in 2020 over last year. Shimadzu, Waters, and Agilent are the main players. Agilent has had a good innings in this segment for about 15 years in India. However, the company has had quality issues with the pharma industry since 2008 and is now not so popular. Its share is being captured by Thermo Fisher and Shimadzu.
Specialty systems are more niche products and did not see much traction in 2020. They may be segmented into:
GPC: Dominated by Waters;
SFC: Leading players are Shimadzu, Waters (Thar); Knauer and Novasep also have some presence;
Ion chromatography: Leading players are Thermo Dynex, and Metrohm;
Preparative chromatography: Leading players are Gilson, and Shimadzu; Novacep, Knauer, Waters, and Agilent also have presence.
The industry is looking for cost effective solutions now that have less operating cost, and yet are robust. Cost of ownership has become a major consideration. With COVID-19 in play, remote controlled serviceable products are being sought, so that dependence on service engineers is minimum. This has impacted the smaller companies as there has been a pressure on margins.
Dual systems are gaining preference, as they offer higher productivity. The Dual LC system contains a dual gradient pump and also a dual injection autosampler. When this is combined with two detectors then essentially there are two UHPLC systems in one and both flow paths are completely independent of each other, so can two things can be done at once.
There is cautious optimism for 2021. Prospects are good, and outlook is bright, yet how the second COVID-19 wave impacts demand is uncertain. JFM 2021 quarter was brisk, and AMJ seems to be promising too.
The global high-performance liquid chromatography (HPLC) market size is projected to reach USD 5.7 billion by 2025 from USD 4.5 billion in 2020, at a CAGR of 4.6 percent, states MarketsandMarkets. The global HPLC market is primarily driven by the high sensitivity and accuracy of the HPLC technique. The ability of HPLC in identifying the drug and monitor diseases and the adoption of hyphenated techniques is further accelerating the growth rate of the market. Moreover, an increase in R&D investments is fueling the HPLC market. The rise in the contract research organizers and the pharmaceutical industries globally may propel the market growth. Furthermore, the rise in the economy of the countries like China and India may boost the market in the coming years. The manufacturers are mostly concentrating on new technologies to help researchers get high-quality analysis that is a significant opportunity for the HPLC market growth. Moreover, the increase in demand for the HPLC technique will boost the market.
HPLC instrument is critical for clinical studies or research. It is required to determine inorganic impurities such as plant or animal proteins existing in pharmaceutical bulk materials or excipients or catalysts. It is also used in the testing of organic compounds. Clinical research involves evaluation studies of the developed drug. This calls for efficient analytical testing instruments like HPLC. Thus, a large market share is witnessed of this application which is followed by the use of HPLC in other application.
Chromatography instruments are equipped with advanced features and functionalities and thus are priced at a premium. The cost of a new instrument differs as per the applications. Chromatography instruments used in the pharmaceutical industry are expensive as they use capillary columns to separate compounds like oxygen, hydrogen, and methane. Small- and medium-sized companies in the biotech and pharmaceutical industries, as well as research and academic institutions, require many such systems in their processes. Hence, the capital cost spent on these systems increases significantly. Moreover, academic research laboratories find it difficult to afford such systems, as they have controlled budgets. The maintenance costs and other indirect expenses result in an overall increase in the total cost of ownership of these instruments. This restricts the adoption of chromatographic accessories and consumables among price-sensitive end users, such as research laboratories, academic institutes, and small companies.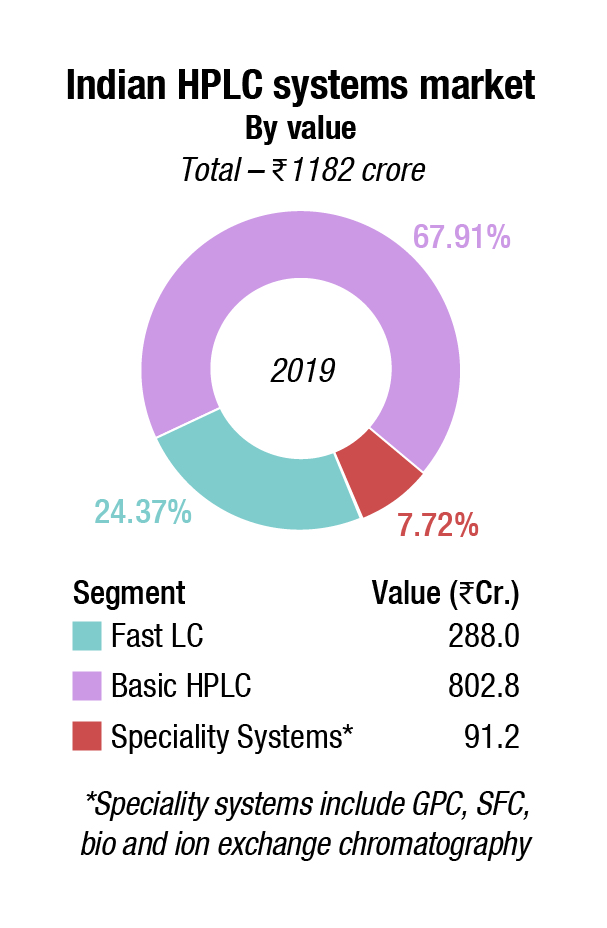 Precipitation, high-resolution ultrafiltration, crystallization, high-pressure refolding, charged ultra-filtration membranes, protein crystallization, capillary electrophoresis, aqueous two-phase extraction, three-phase partitioning, monoliths, and membrane chromatography are some of the prominent techniques that are available as alternatives to chromatography. These techniques can be used as alternatives to column chromatography, which is extensively used to manufacture protein therapeutics, and also for the separation of monoclonal antibodies. Column chromatography employs a minimum of two steps to manufacture protein therapeutics, which increases the cost. Furthermore, column chromatography requires additional steps such as equilibration, wash, elution, regeneration, and sanitization, which escalate the cost even further. On the other hand, alternative techniques require fewer steps and a large volume of antibodies can be separated in a single step. This helps in reducing the manufacturing cost, and also helps in increasing the manufacturing volume. Such benefits provided by alternative techniques are likely to hinder the growth of the chromatography accessories market.
Precipitation, high-resolution ultrafiltration, crystallization, high-pressure refolding, charged ultra-filtration membranes, protein crystallization, capillary electrophoresis, aqueous two-phase extraction, three-phase partitioning, monoliths, and membrane chromatography are some of the prominent techniques that are available as alternatives to chromatography. These techniques can be used as alternatives to column chromatography, which is extensively used to manufacture protein therapeutics, and also for the separation of monoclonal antibodies. Column chromatography employs a minimum of two steps to manufacture protein therapeutics, which increases the cost. Furthermore, column chromatography requires additional steps such as equilibration, wash, elution, regeneration, and sanitization, which escalate the cost even further. On the other hand, alternative techniques require fewer steps and a large volume of antibodies can be separated in a single step. This helps in reducing the manufacturing cost, and also helps in increasing the manufacturing volume. Such benefits provided by alternative techniques are likely to hinder the growth of the chromatography accessories market.
 Advancements and trends in HPLC
Advancements and trends in HPLC
Amit Chopra
Managing Director,
India and South Asia,
Thermo Fisher Scientific
The increasing demand on the pharma and biopharma industry to deliver safe and effective medicines that meet regulatory standards presents a unique set of analytical challenges. High-performance liquid chromatography (HPLC) is one of the most prominent and widely used techniques with extensive applications in analyzing drug samples, quality control, manufacturing complex pharmaceutical drugs and for obtaining faster, and accurate compound separation results. Since its inception, HPLC has undergone technological enhancements in all aspects, including improvements in detectors, advancements in column and pump technologies, precision and automation. These advancements have assisted in conducting high-quality analysis that provide more accurate results in less time while enhancing the overall convenience for the users.
As the industry continues to evolve, there has been a shift in focus of pharma companies toward novel biologic medicines. Advances in columns, stationary phases, detectors, injection devices, as well as the improvements in chromatography methodology, are all leading to an exceeding number of applications, particularly in the pharmaceutical and biopharmaceutical sectors. This has further strengthened the utilization of ultra-high-performance liquid chromatography (UHPLC) applications, particularly for monoclonal antibody characterization, antibody – drug conjugates, quality control or analytical technology in bioprocessing applications. These systems are developed to address the challenges of drug development workflows which can be demanding, require extensive data collection, organization, analysis, accurate results, and fast separation.
With the analytical demands of scientists, working with challenging analytes in complex sample matrices evolve, the implementation of advanced instrumentation and innovations continue to play a pivotal role in meeting the growing needs and in ensuring scientists are able to conduct their work, uninhibited by their instrument capability and capacity.
North America holds the major market share. In 2020, it registered USD 1.26 billion and by 2025, the market is expected to record USD 1.62 billion with a CAGR of 5.2 percent. The increase in R&D investments in the US is the primary reason. The booming pharmaceutical industry in Canada is fueling the market too. The US Food and Drug Administration’s recommendations on the use of chromatographic techniques to ensure proper identity purity, and concentration of pharmaceuticals increase the use of high-performance liquid chromatography techniques. The government’s policies and funding are very supportive.
New technologies are continuously being introduced to improve the efficiency of the analytical laboratory. One area where efficiency could be increased is the analysis of samples at the source. This is particularly important in a range of analytical processes and has resulted in a significant rise in the use of hand-held spectroscopic instrumentation, for example. However, it is not just spectroscopic devices that can be employed for at-source sample analysis. Portable LC systems in particular also offer significant advantages for certain applications.
There is a growing need for scientists to be able to perform analytical measurements at the sample source, instead of collecting samples and returning them to a central laboratory for analysis. This approach can help in terms of reducing the analysis costs but will also have an impact on the timeliness of the data and potentially its integrity as well if the sample degrades. Historically, this has been difficult to achieve due to the size, cost, and complexity of the instrumentation that is required to perform the analysis. Recent advances in fluidics and LED technology have facilitated the design of HPLC systems small enough to be fully portable rather than luggable. This allows samples to be analyzed in situ, bringing substantial benefits; however, it is important that in doing so there is no compromise in the quality of the data that is being produced. There are a range of such instruments that are currently available.
The instrumentation has been used in a variety of different applications to great effect. The following applications highlight the portability and applicability of this type of instrument.
Hemoglobins. Hemoglobin is probably the most widely analyzed protein in the world. About 7 percent of the human race is carriers of a gene for a significant hemoglobinopathy such as sickle cell disease. In parts of sub-Saharan Africa the incidence of carriers is as high as 35 percent. The version of the SmartLC used for haemoglobin analysis is called SmartLife LC, and uses a detector wavelength of 415 nm. Six units are now in use in sub-Saharan Africa. An obvious use of a portable HPLC is its use in the field for analysis of haemoglobin variants in remote regions. This can involve either a single drop of blood (4 µl of whole blood suffices for 50 replicate analyses) or archived samples in the form of dried blood spots. The out-of-pocket expense for the reagents and HPLC column usage is about USD 1 per analysis.
Cannabinoids. This is one application where analysis in the field could be helpful. An example would be the analysis of extracts of cannabis from different locations at a farm to get more detail about differences in cannabinoid composition in the different places. This version of the machine is called SmartCanna LC, with a detector wavelength of 280 nm.
In addition to the wavelength of the detector module, the most common modification to date has involved the controller. It is somewhat cumbersome to enter data directly on the tablet that comes with the unit; a USB or Bluetooth keyboard and mouse make the task easy. A PC can be substituted for the tablet as the controller, in which case Clarity is then loaded onto the computer and the license transferred to it. The default mobile phase bottles have a 500-ml volume and fit inside the case. There is no reason why they have to be inside the case during operation, and some customers have ordered 2-litre. bottles for the purpose instead. They must be carried separately when the unit is moved, a minor inconvenience. Add-ons such as a sample carousel and automatic sample injector would certainly compromise the portability of the device, as mentioned earlier, and would require operation using line current rather than the battery. However, if the machine is to remain in one location for an extended period, then such modifications would increase its capabilities to the point that they approach those of a conventional HPLC.
Portable HPLC machines have not been available long enough to have entered markets, which would truly exploit their portability. There are numerous application areas that would benefit from the technology, and move chromatography away from a laboratory based activity to one which is more inclusive of the population. One such niche would be analysis of water samples or fruit in the field in real time, rather than having to transport them back to a central lab. The results could inform the locations being sampled. As the technology in this area develops through the use of lighter components and higher-functioning separation and detection technology, the possibility of moving chromatographic analysis into the world at large becomes a potential reality. This would start to move the technology toward a future where separation science is seen as a means of ensuring a better lifestyle.
 HPLC – The foundation for clinical diagnostics
HPLC – The foundation for clinical diagnostics
Bhaumik Trivedi
Sr Application Scientist,
Shimadzu Analytical (India) Pvt. Ltd.
Ever since the 1970s liquid chromatography, gas chromatography and thin layer chromatography have provided analytical solutions in diagnostics laboratories. HPLC (high-performance liquid chromatography) is the advanced types of chromatography. It is given prominent importance due to its attributes like high sensitivity, i.e., the ability to evaluate samples of very minute concentrations, detect precisely chemically similar molecules like monoamines and ability to identify compounds with complex chemistry. This is possible in HPLC due to efficient separation under pressure over a large surface area.
The HPLC system is also connected to detectors like UV-visible, fluorescence, mass spectrometers, electrochemical detectors, etc.
HPLC system is a mandatory tool in most of the labs. The fields of medical, biological, chemical, biochemical, phytochemicals (plant chemical research) it finds its application to analyze and quantify the molecules. Components with complex chemistry and properties are easily distinguished by this technique. Due to the principle of separation in HPLC, similar molecules get separated and hence their detection, identification, and quantification become easier.
HPLC has played a significant role in clinical laboratories for separation and quantitation of bio markers in different body fluids. Many disorders related to body metabolism, those related to endocrine and exocrine gland secretion, alteration in body fluids are diagnosed by HPLC analysis of concerned fluids.
For example, estimation of metabolites of purines, pyrimidines, or other metabolites from plasma, cerebrospinal fluid, and urine samples in patients. Estimation of corticoids from plasma in disorders of the adrenal gland, which secretes an endocrine hormone. Because of the time factor, most of the diagnostic methods are replaced by ELISA, electrophoresis, and RIA methods. But still, for any new or rare problem, the HPLC method is preferred to pinpoint the cause of disorders (i.e., any change in some biochemistry).
Today HPLC has made a cutting edge over other methods such as immunoassay for routine estimation of vitamins, hormones and other biomarkers. The cost of such advanced analytical techniques are prohibitive but due to the advantages of ultrahigh sensitivity and capability to handle heavy workloads the returns on investment are recovered easily by clinical laboratories through high profits, higher confidence and efficient services to patients.
How the latest HPLC advances are helping analytical testing laboratories achieve optimal productivity. Routine analysis laboratories play a vital role in safeguarding integrity across a wide range of sectors, from food and beverage to forensic and pharmaceutical. With product safety and reliable decision-making on the line, these laboratories must balance productivity with the highest standards of analytical accuracy. Yet, for laboratories operating at or near capacity, or planning to grow their market share, scaling-up workflows without compromising on quality can be challenging.
Liquid chromatography technologies, including HPLC and ultra-high performance liquid chromatography (UHPLC) platforms, support a large proportion of routine analysis workflows. However, these systems can be a major source of inefficiency and serve to limit operational productivity. In particular, issues with the adaptability and resilience of aging LC equipment can be highly disruptive to workflows, making method transfer complex and increasing the need for time-consuming manual steps. Poor platform flexibility and robustness can also leave processes vulnerable to human error, resulting in the need for additional efforts to document out of specification results.
Routine analysis laboratories may face several potential productivity challenges with regards to their LC workflows. One of the most common sources of inefficiency is the process of transferring LC methods between instruments, where unforeseen complications and delays can divert time away from testing and have a major impact on productivity. This is true not only when transferring methods between laboratories, such as from analytical development to quality control (QC) settings, or when outsourcing to contract laboratories, but also when scaling-up methods across multiple instruments within the same laboratory or transferring workflows from legacy to new systems.
The ease and speed of method transfer often depends upon several factors, including the robustness of the method to be transferred, as well as the instrumental deviations of the systems involved. Here, the technical characteristics of the platform, such as gradient delay volume, pump mixing mode, hydrodynamic behavior, choice of column, and eluent thermostatic options, can all affect critical performance outcomes, such as peak resolution or retention times. The complexity of method transfer is also determined in large part by the requirements of the analytical outcome and defined limits of acceptable deviation from the original system. Ideally, method transfer should occur with no method modifications to avoid spending large amounts of time and resources re-validating methods.
 Dr Shaloo Kapoor
Dr Shaloo Kapoor
Chief Pathologist and Sr DGM,
SRL Diagnostics Ltd,
Fortis Escorts Heart Institute, New Delhi
“HPLC is complex method based on column chromatography with wide range of applications in diagnostic labs.
The emerging hyphenated techniques incorporate chromatography – mass spectrometry (LC-MS) combining the physical separation properties of liquid chromatography (or HPLC) with the mass properties of MS (mass spectrometry). More widely adoption needs development of user friendly analyzers and their smooth interfacing with LIS. Innovations in LOC include micro and nano fluidics and SFC chip. Technology enhancements in HPLC are likely to increase throughput, gain more confidence in results and drive down cost per sample.”
Other important causes of low productivity often stem from small yet critical oversights during routine laboratory processes, which can have major consequences for operational success. For example, poorly-maintained eluent levels will cause sequences to stop mid-run if there is insufficient solvent to complete an analytical sequence, resulting in the loss of samples, sample re-analysis, and if working in a regulated environment, documentation for regulatory authorities. Compromised HPLC systems are also a key source of laboratory inefficiency, leading to unreliable and out-of-specification results that take valuable time out of routine workflows and prevent laboratories from achieving optimal productivity. Equally, recognizing these performance issues too late can result in extended instrument downtime due to unscheduled maintenance.
With productivity now a key focus point for analysis laboratories across all sectors, instrument vendors have responded by developing HPLC technologies designed to overcome these challenges.
One of the main reasons why method transfer can be so time-consuming is that success is often not defined by a single instrumental parameter. Factors such as gradient delay volumes, column thermostatting, and system dispersion effects all play important roles. Poor system configurability through rigid instrument design can, therefore, be a major stumbling block. Fortunately, recent years have seen a heightened focus on increasingly flexible HPLC technologies that can help laboratories streamline method transfer through the application of highly customizable parameters.
Some modern instruments now offer a wide range of customizable features to facilitate the precise replication of existing methods. Highly flexible platforms, such as the Thermo Scientific Vanquish Core HPLC system, for example, allow gradient delay volumes to be seamlessly adjusted to match the analyte retention time and separation profiles of other instruments. Other innovations, such as a choice of still-air and forced-air thermostatting techniques within the same instrument, can help analysts more easily mimic legacy HPLC systems to achieve the desired results faster.
Another key issue associated with method transfer relates to system dispersion effects. While lower system dispersion benefits overall separation power, it can lead to undesirable peak shapes when samples with high organic content are injected. Flexible systems that use custom injection programs to perform in-needle dilutions have enabled analysts to inject high organic samples, while maintaining exceptional chromatographic efficiency. As such, custom injection programs are increasingly seen as a powerful tool when it comes to rapidly transferring methods between systems.
While increased platform flexibility is streamlining method transfer workflows, other improvements in productivity are being brought about by advances in intelligent informatics and instrument design.
Among the most valuable productivity gains are those achieved by integrating HPLC instrumentation with laboratory informatics software, including chromatography data systems (CDS). By allowing analysts to set-up sequences, monitor runs, and access data remotely using a CDS, these software solutions are helping teams run more samples unattended and work more efficiently to deliver high-quality results first-time around. In recent years, CDS platforms have become increasingly sophisticated, with some systems now supporting electronic workflow procedures designed for fast, error-free sequence and method set-up. Other features, such as dedicated panels for instrument control and smart tools to streamline data processing and reporting, are empowering analysts to work faster and boost productivity, while ensuring all data is managed in full compliance with regulatory requirements.
Innovative productivity tools are also being built into instruments themselves. For example, some modern HPLC platforms can monitor critical environmental parameters, such as eluent and waste solvent levels, providing automatic warnings to notify if there is insufficient eluent to complete sequences and automatically recording eluent bottle refill events in the audit trail. These simple yet effective features are designed to ensure instruments never run dry and waste will not run over, enabling laboratories to minimize the potential for human error, avoid lost time, and ultimately run large sample sets with confidence.
Recent years have also seen the development of HPLC instruments that can monitor operational readiness through routine and automated system health checks. As a result, the early tell-tale signs of poor performance can be flagged in good time, ensuring any potential issues are resolved early, and significantly reducing unscheduled downtime or the need to re-run samples. Some of the most advanced LC instruments also incorporate intuitive diagnostic tools to quickly identify and troubleshoot issues. These systems can even display step-by-step maintenance videos on intuitive touch-screen displays, helping analysts quickly remedy issues and get workflows back online and running again faster.
Productivity is a key challenge for analytical laboratories across a variety of sectors and the pharmaceutical industry is no exception. For contract development and manufacturing organizations (CDMOs), who work with a wide range of customers, diversity in analytical capability is an important consideration.
This was the situation facing a global CDMO looking to increase the flexibility and efficiency of its LC systems in order to enhance its capabilities and expand its customer base. A key factor in this decision was the need for a LC platform that would simplify and accelerate method transfer workflows. Design features, such as active solvent pre-heating, were critical. This allowed the laboratory to standardize analytical performance through enhanced unit-to-unit thermal consistency, while the ability to use tool-free fluidic connections ensured full compatibility with any separation column. Other innovations, including built-in adjustable gradient delay volume control and multiple column heating modes, allowed teams to fine-tune method parameters to achieve the highest levels of reproducibility, further supporting faster method transfer.
Deployment of the new UHPLC platforms at its sites across the globe helped the organization expand its assays, such as released glycan analysis, peptide mapping and monitoring, as well as purity and ratio analysis. By increasing operational flexibility, these solutions played a critical role in boosting efficiency and productivity, while maintaining the highest standards of analytical excellence.
As workloads increase and businesses look to expand, analytical laboratories across multiple sectors are turning to the latest HPLC technologies to drive improvements in productivity. Thanks to modern HPLC solutions, forward-thinking laboratories are streamlining the movement of both analysts and samples to achieve substantial gains in capacity and efficiency.












