Urinalysis Instruments and Reagents
Unlocking the power of urinalysis through automation

Recent advancements in automation have revolutionized urinalysis, unlocked its full potential, and transformed the way it is performed.
Urinalysis, the analysis of urine samples, has long been a cornerstone of diagnostic medicine, providing valuable insights into a patient’s health status. However, traditional manual methods of urinalysis are labor-intensive, time-consuming, and prone to human error. The emergence of automation in the field of urinalysis has brought about a paradigm shift, unlocking its full power and potential. By harnessing advanced technologies and innovative systems, automation is revolutionizing the way urinalysis is performed, offering unprecedented efficiency, accuracy, and data analysis capabilities.
Automation in urinalysis encompasses a range of advancements, including automated sample handling, processing, and analysis. Sample preparation, a critical step in urinalysis, can now be performed with minimal human intervention, ensuring consistent and reliable results. Automated systems can handle large volumes of samples, track them throughout the testing process, and reduce the risk of sample mix-ups or contamination. This improved sample management not only saves time but also enhances accuracy and traceability.
The introduction of automated analyzers specifically designed for urinalysis has further enhanced the power of this diagnostic tool. These analyzers are equipped with sophisticated sensors, robotic arms, and advanced algorithms that can perform a wide array of tests on urine samples. From measuring physical and chemical parameters to identifying microscopic elements, these automated analyzers provide comprehensive and rapid results. The high-throughput capabilities of these systems allow laboratories to process many samples in a shorter time frame, enabling faster diagnosis and treatment decisions.
Furthermore, automation in urinalysis improves data management and analysis. Integrated workflow systems connect different components of the urinalysis process, allowing seamless data exchange between instruments and systems. This integration enhances the accessibility and traceability of results, facilitates remote monitoring and reporting, and enables data-driven decision making in healthcare settings. The ability to integrate urinalysis data with electronic health records (EHRs) or data analytics platforms paves the way for advanced analytics, population health studies, and personalized healthcare strategies.
The increasing demand for robotics and the evolution of personalized medicine are expected to further propel market growth in the long term.
Indian market
The Indian market for urinalysis analyzers and reagents in 2022 is estimated at ₹230 crore. Reagents continue to dominate with an 81-percent share, valued at ₹186 crore. Most of the instruments in this segment are closed systems.
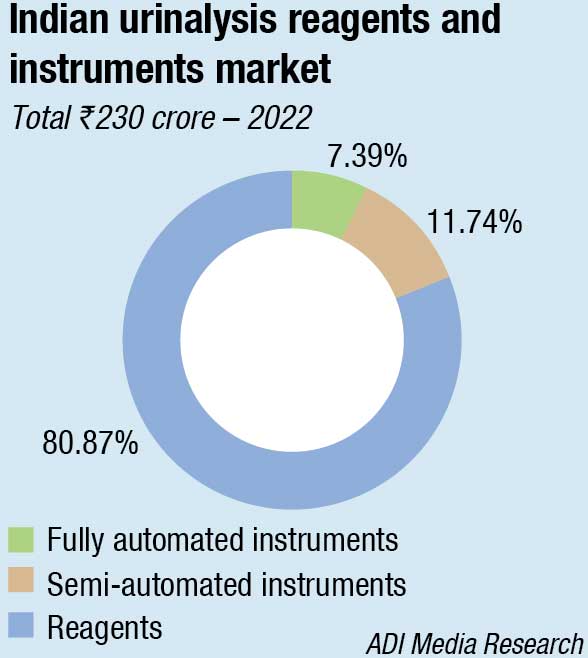
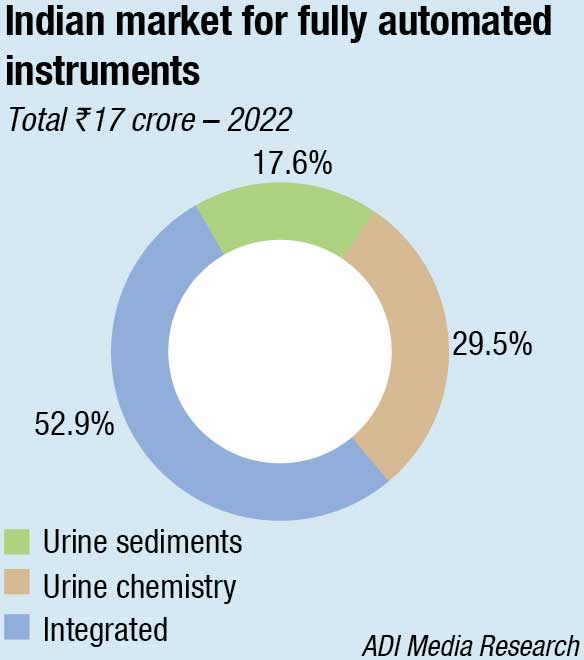
The fully automated analyzers segment is estimated at ₹17 crore in 2022. There is a distinct shift toward automation as it decreases observer-associated variation and offers prompt competitive results when standardized for screening away from the borderline cutoffs. Also, the documentation support required by NABL is possible only with fully automated instruments. The urine chemistry instruments are now more popular, with their counterpart, the urine sediment analyser segment being catered to by a handful of companies. The integrated instruments continue to dominate, with the medium-sized diagnostic centres showing a strong preference for them. The larger diagnostic centres are finding success with the rental instruments, with more and more companies giving in to placements.
AI is being increasingly applied in urinalysis. An AI module is developed in the WebApp software, where it merges all available algorithms and allows the use of tailored rules to provide presumptive results automatically, which can be accepted or overruled by the lab technician. This work describes how to design the tailored module, perform its validation on large data collections of urine and check its performance in routine practice 3 months after implementation. Offered at an average unit price for ₹900,000, AI-based instruments are not finding many takers in the Indian market.
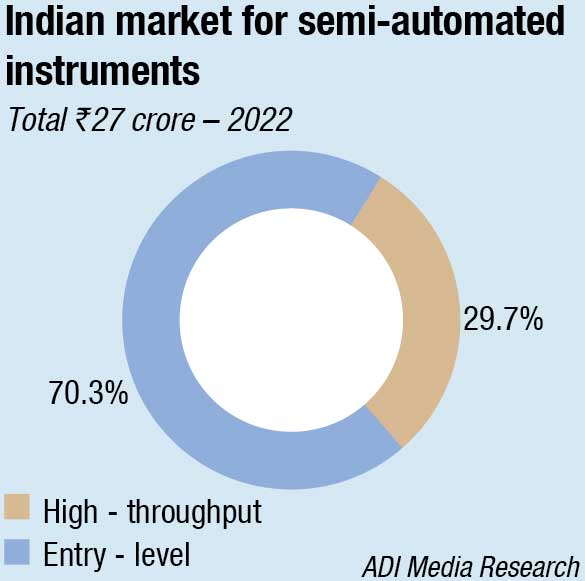
The semi-automated instruments category, amounting to ₹27 crore, were the reason that the market in 2022 grew, having witnessed a 23-percent increase. The segment had witnessed a 40-percent increase in 2021 over 2020 too. The entry-level instruments continue to constitute 70 percent of the market by value.
Many companies that were earlier importing instruments as fully finished products are now importing them in SKD condition, since the new regulation introduced in October 2022 is moving toward ensuring that value added in India is higher. This is also increasing the size of the Indian market for instruments.
Global market
The global urinalysis market was valued USD 3.9 billion in 2022 and is expected to reach USD 5.7 billion by 2027 at a CAGR of 8.1 percent, according to MarketsandMarkets. The urinalysis market has witnessed substantial progress and broadened its horizons in recent years, primarily driven by significant advancements and expansions in technology and testing methodologies.
Factors, such as the rise in the geriatric population and its associated disorders, the ease of conducting urinalysis tests, and the availability of comprehensive results, have generated a significant demand for urinalysis, thereby driving the global market for urinalysis testing products.
In addition, the incidents of increase in kidney diseases and urinary tract infections, particularly in the geriatric population, and the growing occurrence of chronic diseases like diabetes and hypertension has contributed to the increase in the demand for urinalysis testing products across the globe.
In February 2023, the World Health Organization (WHO) released data indicating that the number of individuals, aged 60 and above affected by urinary tract infections and asymptomatic bacteriuria, is projected to increase from 1 billion in 2020 to 1.4 billion by 2050. Differentiating between the two conditions can be challenging. As per the National Institutes of Health (NIH), the prevalence of UTIs is estimated to be around 10 percent in men and 20 percent in women aged 80 and above.
Additionally, the rising demand for point-of-care (POC) diagnostic testing has driven the development of innovative urinalysis testing equipment. These advanced devices provide quick and accurate results, making them highly suitable for emergency rooms, outpatient clinics, and other healthcare settings.
The market faces a significant hurdle in the form of the expensive nature of automated urinalysis systems and other diagnostic testing equipment. Furthermore, the lack of proficient professionals to perform diagnostic tests, particularly in developing countries, is a key factor impeding market growth. Another notable constraint is the potential for inaccurate results obtained from certain urinalysis tests, which can lead to misdiagnosis and inappropriate treatment. Additionally, the absence of standardized urinalysis testing methods across different laboratories makes it challenging to compare results, further complicating the situation.
Regional insights
North America accounts for the major urinalysis market. The reasons are many, but the most important is the significant healthcare expenditure. Rise in the number of patients with chronic renal diseases, hypertension, and diabetes is also a reason for the region holding the largest chunk of the market share.
The European market is anticipated to exhibit a robust revenue growth rate throughout the forecast period. This growth can be attributed to the favorable reimbursement policies, government guidelines and regulations, and the advanced healthcare infrastructure that exists in the region.
The Asia-Pacific market is anticipated to demonstrate a significant revenue growth rate throughout the forecast period, driven by the rapid advancement of the healthcare industry in the region and increased investments from key market players.
The key players operating in the global urinalysis equipment market are Siemens Healthineers AG, Roche Diagnostics Ltd., Cardinal Health Inc., Abbott Laboratories, Danaher Corporation, Beckman Coulter Inc., Sysmex Corporation, ARKRAY Inc., ACON Laboratories Inc., Bio-Rad Laboratories Inc., URIT Medical Electronic Group Co. Ltd., 77 Elektronika Kft, Thermo Fisher Scientific Inc., Teco Diagnostics, Mindray Medical International Limited, and Quidel Corporation.
|
Major Players Fully automated instruments & reagents 2022 |
||||
| Urine chemistry | Siemens, Beckman Coulter, Tranasia, Sysmex, Agappe, Rapha, Arkray, and Roche. | |||
| Urine sediments | Siemens, 77 Electronika (Suyog), Sysmex, Dirui, Roche, URIT, Laura, and Beckman Coulter. | |||
| Integrated | Sysmex, Transasia, Rapha, Beckman Coulter, and Dirui. | |||
| Semi-automated instruments & reagents (incl. strips)
2022 |
||||
| Tier I | Tier II | Tier III | ||
| Siemens and Transasia | Agappe, Rapid and Tulip | Iris (Beckman Coulter), Rapha, AVE, China (marketed by Matrix Chennai), Abbott, Dirui and Mission. | ||
| *Vendors are placed in different tiers on the basis of their sales contribution to the overall revenues of the Indian urinalysis market.
ADI Media Research |
||||
Automation in urinalysis
Over time, the field of urinalysis has experienced significant progress, leading to increased automation. Automated equipment, such as refractometers and osmometers, have been developed to facilitate the precise determination of physical and chemical properties in urine samples. For microscopic analysis of particles, technologies like flow cytometry, fluorescence flow cytometry, impedance, and light scattering are employed. These advanced techniques enable the detection and characterization of different elements present in urine samples.
Automation has transformed the field of urinalysis, offering significant benefits in terms of efficiency, accuracy, and data analysis capabilities. Automated sample handling, processing, and analysis have revolutionized the way urinalysis is performed, ensuring consistent and reliable results, while saving time.
Automated analyzers, equipped with advanced sensors and algorithms, provide comprehensive and rapid results, enabling faster diagnosis and treatment decisions. The integration of urinalysis data with electronic health records and data analytics platforms allows for advanced analytics and personalized healthcare strategies.
Automation brings several advantages, including improved productivity, reduced errors during pre-analytical, analytical, and post-analytical stages, and faster processing times. However, it also has some drawbacks, such as reduced employment opportunities for laboratory professionals and challenges in analyzing pathological samples, where automation may exhibit suboptimal analytical performance.
Evaluating color correction algorithms for automated interpretation of urinalysis dipsticks with low-cost image sensors. Aimed to assess color correction techniques and reduce variability in color interpretation for urinalysis dipstick results in using a low-cost imager. Visual color readings in medical diagnostic tools like urinalysis dipsticks can be subjective and influenced by various factors. The researchers explored the use of low-cost camera modules for objective color readouts but needed to apply color correction to account for deviations. Three correction methods were compared, and the root polynomial regression showed the best performance in reducing color differences.
Urine dipstick analysis on automated platform. An automated urinalysis dipstick method was evaluated by comparing its results with quantitative tests for creatinine and albumin on a clinical chemistry platform.
The semi-quantitative dipstick test provides a valuable tool for early detection and prevention of renal damage and associated cardiovascular risks. It serves as a complementary method to the quantitative test and is particularly useful for population-wide screening programs. However, the quantitative method should be used to confirm ACR values in cases of abnormal results.
Sysmex Corporation launched two models in the last one year, the UF-1500 fully automated urine particle analyzer. The UF-1500 maintains the same high levels of functionality, and usability, by using the same advanced fluorescence flow cytometry technology of the UF-5000. And the UN-series that automates the lab’s urinalysis work area according to workflow preferences. With modular components to mix and match, a complete solution can be tailored that is scalable to current and future needs, providing a perfect fit. The series offers urine flow cytometers UF-5000 and UF-4000; UC-3500 is a fully automated chemistry analyzer; for smaller workloads, the semi-automated chemistry analyzer UC-1000; digital imaging unit, the UD-10; optional decapping module TH-11 automates urinalysis workflow seamlessly right from the point of sample entry, and U-WAM takes care of the data and sample management. The analyzer uses Sysmex’s proprietary fluorescence flow cytometry technology to provide rapid and accurate results for urine analysis.
Advancement in urinalysis
 Nilesh Kadu
Nilesh Kadu
Dy. Product Manager (Diabetes Management & Urine Analysis),
Transasia Bio-Medicals Ltd.
Urinalysis plays a pivotal role in diagnosing various medical conditions and monitoring patient health. Over the years, significant advancements have been made in urinalysis instruments and reagents, leading to improved accuracy, efficiency, and diagnostic capabilities.
Enhanced accuracy
Modern urinalysis instruments leverage state-of-the-art technologies to deliver highly accurate results. Automated analyzers utilize advanced algorithms and optical systems to precisely measure various parameters, including urine chemistry, sediment analysis, and microscopic elements. The integration of intelligent software helps minimize human error and ensures reliable and reproducible results, contributing to more accurate diagnoses.
Comprehensive test panels
Urinalysis instruments now offer expanded test panels, allowing simultaneous analysis of multiple analytes in a single sample. These comprehensive panels encompass parameters such as glucose, proteins, ketones, pH, nitrites, leukocytes, and many more. This broader range of analytes provides a more holistic assessment of a patient’s urinary health, aiding in the detection and monitoring of various conditions like urinary tract infections, kidney diseases, diabetes, and liver disorders.
Advanced sediment analysis
In the case of digital microscopy based sediment analyzers, the availability of high resolution sediment images for review as well as the option to save and store them has remarkably advanced the scope of testing. This automated approach improves the detection of abnormal cell types, crystals, casts, and other sediments, enabling clinicians to make more precise diagnostic decisions and identify potential kidney or urinary tract disorders.
User-friendly interfaces
Intuitive touchscreen displays, step-by-step guidance, and customizable workflows enhance user convenience and efficiency, enabling faster adoption and reducing the learning curve for laboratory personnel.
Quality reagents
Alongside instrument advancements, improvements in urinalysis reagents have played a vital role in enhancing diagnostic accuracy. Reagents now come with improved stability, extended shelf life, and enhanced analytical specificity. Moreover, reagents are formulated to work seamlessly with specific instrument platforms, ensuring compatibility and reducing potential errors.
Iris urine analyzer – The modern tool for urinalysis from Beckman Coulter. Iris iChemVelocity urine analyzer is an automated device that offers advanced features and technology to streamline and enhance the urinalysis process. It utilizes state-of-the-art optical systems, image recognition algorithms, and automation to analyze urine samples rapidly and accurately.
The automated system can process many urine samples in a short period, reducing turnaround time and enabling prompt diagnosis and treatment decisions. The automation offered by the Iris urine analyzer reduces the risk of human error and variability in test results.
By integrating advanced technology and automation, the Iris urine analyzer has revolutionized the field of urinalysis, providing healthcare providers with a reliable and efficient tool for diagnosing various conditions and monitoring patient health.
For diagnostic labs, three things continue to be hot issues regardless of instrumentation, field of analysis, and subspecialty. Number one is automation in workflow, having something that is robust for high volume, low staffing. Number two is the need for fit of an instrument line. In other words, there is a core lab urinalysis instrument that is ideal and yet there are smaller clinics, hospitals, and all kinds of sites that can benefit from the same technology but in a differently configured unit. Number three is understanding if we are extracting the optimum clinical information from the analysis in fields like hematology and urinalysis.
Urinalysis- Automation is leading the way
 Shobhit Jain
Shobhit Jain
Sr. Manager, Product – Clinical Chemistry, Urinalysis, Marketing Events and Branding Marketing,
Sysmex India Pvt Ltd
Urine analysis is comprising of physical, routine and microscopic examination. It is an integral part of clinical laboratories and one of the most performed diagnostic tests. This simple, noninvasive test is potent screening tool for clinicians, which can provide as much as possible information on the status of renal, urological, infection and metabolic function in the body. Apart from routine screening, it may also be helpful in uncovering status of disease that may not be showing significant signs or symptoms.
A lot of vital information can be obtained through urine examination, still its clinical utility is affected to a great extent by limitation of its conventional manual methods including bio-chemical and microscopy. Biochemical analysis is improved with reagent strips, but challenge remained in microscopic part. Other challenges like non standardization of process, person to person variation, centrifugation and slide preparation needed a whole automated system in place with minimum human involvement and maximize automation.
In last decade there were paradigm shift in laboratory operations and now automation is leading the way for urinalysis testing. Technological advancement in biochemical analysis and microscopic examination in urinalysis has help to set and standardize process. Automation has changed the way of testing, reporting and documentation of laboratories with improved quality and lowered TAT. Some of the key features of automation in urinalysis are standardization, quality control, added clinical values, data management, accuracy, and reproducibility.
Automation in urinalysis can be further categorized in bio-chemical analysis and particle analysis (Microscopy). In bio-chemical analysis, CMOS sensor has played a great role in identifying strip pad position, optimizing detection area, correction of abnormal coloration. This cutting-edge technology has eliminated all possible errors due to manual process. Similarly, for particle analysis, fluorescence flow cytometry technology has great advantage as it can recognize specific and unique properties of different particles by size, labeling DNA, complexity, and specific features, and so determining the particle type. Also, high accuracy of bacteria counts and, accurate differentiation in RBC morphology (isomorphic and dysmorphic) is helping in faster diagnosis and better patient care. Looking at the facts it can be concluded that future of urinalysis lies in automation.
The clinician needs to be able to make the best decision for their patient in terms of test orders. Reflex testing can help laboratories with optimizing utilization, which can significantly impact turnaround times. If a laboratory is performing unnecessary testing – testing that would not have been performed if governed by optimal reflex test criteria – then it is spending time and resources that could be used for other, more value-added tasks.














