


Chinese vaccine developer CanSino Biologics Inc said on Monday that its potential Covid-19 vaccine using the messenger RNA (mRNA) technology has been approved by China’s medical...



The Southeast Minnesota Capital Fund is a small but increasingly popular investment firm focused on funding good ideas in the Rochester startup scene. While the vast...

Sweden recommended a fourth Covid-19 vaccine dose to people 65 and over as well as those living in nursing homes or getting home care, authorities said...



China has sent more than 10,000 health workers from across the country to Shanghai, including 2,000 military medical staff, as it struggles to stamp out a...



As governments prepare to live with Covid-19, some are questioning how much to rely on drugmakers to adapt vaccines to ward off future virus variants amid...

The World Health Organization said on Saturday it has suspended supply through United Nations agencies of Covid-19 vaccine Covaxin, produced by India’s Bharat Biotech, to allow...

A new mutant of Covid-19 called ‘XE’ appears to be about ten per cent more transmissible than the BA.2 sub-variant of Omicron, as per the WHO....



Seoul National University Bundang Hospital (SNUBH) said that it would build a platform for researching and developing medical artificial intelligence startups with GE Healthcare Korea. SNUBH...



Mexican Foreign Minister Marcelo Ebrard has begun talks to receive medical technology from Indian companies to produce vaccines and cancer drugs in Mexico on Wednesday, the...

The lawsuit in a Seattle court by a US start-up may not immediately impact the launch of Pune-based Gennova Biopharmaceuticals’ mRNA vaccine HGCO19 once it receives...
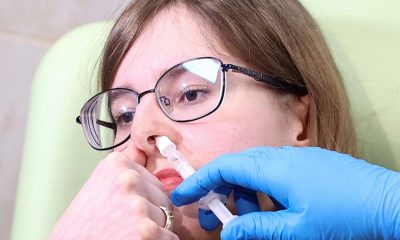

The Russian Health Ministry has registered the nasal version of its vaccine against coronavirus – Sputnik V. It is the world’s first nasal vaccine against Covid-19....



The U.S. Food and Drug Administration on Friday approved expanded use of Yescarta, a cell therapy made by Gilead Sciences Inc’s (GILD.O) Kite unit, as a...



Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) has given approval to Shockwave Medical’s Shockwave C² Coronary Intravascular Lithotripsy (IVL) Catheter. The regulatory approval was supported by...



A phase 2 clinical trial evaluating various additional Covid-19 booster shots has begun enrolling adult participants in the United States, the U.S. National Institutes of Health...



French healthcare group Sanofi said it had priced a first sustainability-linked bond issue that will be indexed on access to medicines, and worth 1.5 billion euros...
Copyright © 2024 Medical Buyer