Company News
Mainz Biomed reports full year 2023 financial results

Mainz Biomed N.V. announced financial results for the fiscal year ended December 31, 2023.
“This past fiscal year represented an important period of progress for the Company highlighted by reporting results from our groundbreaking ColoFuture and eAArly DETECT studies which demonstrated extraordinary statistical significance for colorectal cancer (CRC) detection and advanced adenomas, a type of pre-cancerous polyp which are considered precursors to colorectal cancer,” commented Guido Baechler, Chief Executive Officer of Mainz Biomed. “This outstanding outcome positions our much-anticipated ReconAAsense pivotal FDA PMA study to be optimally designed to evaluate a best-in-class self-administered CRC screening tool which if successful, will be the gold standard stool-based diagnostic for this deadly disease. As we make final preparations to initiate ReconAAsense’s patient enrollment, which remains on track to begin later this year, we will continue expanding the international commercialization of ColoAlert®, our highly efficacious and easy-to-use DNA-based detection test for CRC and advance the balance of our product development pipeline.”
Key 2023 accomplishments and recent highlights
- Announced groundbreaking results from ColoFuture (Europe) and eAArly DETECT (U.S.) clinical studies evaluating the potential to include novel gene expression (mRNA) biomarkers into a next generation version of Mainz BioMed’s CRC screening test.
- ColoFuture reported sensitivity for CRC of 94% with specificity of 97% and advanced adenoma (AA) sensitivity of 80%.
- eAArly DETECT reported sensitivity for colorectal cancer of 97% with a specificity of 97% and a sensitivity for advanced adenoma of 82%.
- Final preparations for Pivotal FDA PMA clinical trial (ReconAAsense) evaluating gold standard self-administered CRC test – on track to commence patient enrollment in 2024.
- Established partnership with Trusted Health Advisors, a market leader in supporting the U.S. launches and management of commercial programs for diagnostic products – collaboration led by Jay Wohlgemuth, former Chief Medical & Scientific Officer of Quest Diagnostics.
- Expanded international commercialization of ColoAlert®, a highly efficacious and easy-to-use DNA-based detection test for CRC being sold via Company’s unique business model of marketing products via partnerships with third-party laboratories versus the traditional methodology of operating a single facility.
- Established commercial operations in major markets including Spain, Poland, Romania, Portugal, Israel, and the UK.
The company opened its own physician-led laboratory, ‘European Oncology Lab’ (EOL), thereby offering ColoAlert® as a laboratory medical service directly to physicians and patients, granting potential for further expansion into Germany’s private health insurance segment where the DNA-based test already receives reimbursement.
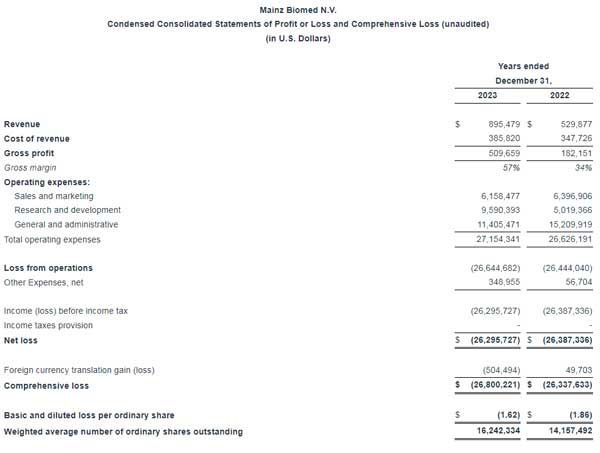
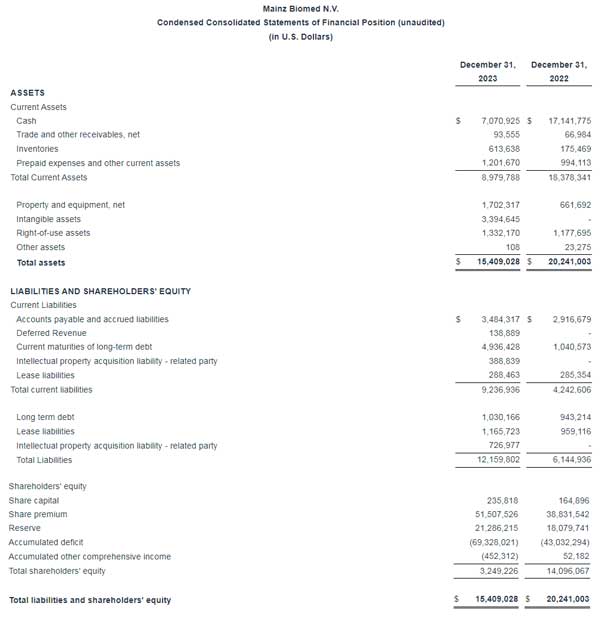
Condensed consolidated financial statements (unaudited):
MB Bureau












