Immunochemistry Instruments and Reagents
Technology Is Driving The Immunochemistry Analyzers Market
With continuous innovation and breakthroughs, testing has taken a new leap. However, with demand slowing, resulting in stiff competition among players, a high number of collaborations and acquisitions are predicted.
The medical care we receive has improved greatly over the years, thanks to technological advancements that allow for earlier and accurate diagnoses that lead to more effective treatments. One such example is the advancement of immunochemistry analyzers that helps to diagnose diseases and other abnormalities within the human system, as well as help confirm the health status. During the past two decades, a wide range of analyzers has been developed to provide the quantitative, semiquantitative, or qualitative detection of analytes. The precise early-stage detection of analytes is an essential requirement for all bioanalytical settings to effectively monitor and manage the quality of biopharmaceutical drugs, foods, and environment. It is even more critical to effectively diagnose, monitor, and manage the patients’ health. Considering the prominent role that immunochemistry analyzers play in the clinical decision-making, they are indispensable for healthcare settings.
Tremendous advances in immunoassay formats, bioanalytical platforms, immunoanalytical systems, and complementary technologies have led to various emerging immunochemistry technologies. Initially, most of the analyzers, developed several decades ago, were based on radioimmunoassay (RIA) and enzyme-linked immunosorbent assay (ELISA). Owing to extremely high sensitivity, specificity, precision, and throughput of ELISA, this format has served as the gold standard for a plethora of analytes and the last two decades have seen tremendous innovation in ELISA technology. Automated assays are equipped with robotic workstations while the microtiter plate (MTP) readers have also improved drastically in terms of technology, features, and cost-effectiveness. Advanced bioengineering, microfluidic, and biosensor technologies have further led to the development of prominent immunoanalytical systems including surface plasmon resonance (SPR)-based instruments for label-free immunoassays. These systems have become the global standard for developing rapid analyzers for the detection of analytes and screening of immunological components based on the determination of biomolecular interactions.
The advent of microfluidics started the quest for novel immunochemistry platforms and formats, which enable rapid procedures using a minimal volume of reagents. An example is the Optimiser ELISA which involves the conversion of the conventional 96-well MTP-based ELISA into microfluidic ELISA. The analysis time is only a few minutes as this hybrid system employs a significantly reduced number of steps and a microfluidic immunoassay protocol. Similarly, a large number of prospective lab-on-a-chip (LOC) technologies and formats have been developed toward the development of various microfluidic devices. The most widely used microfluidic device format is the lateral flow immunoassay (LFIA). Although most of the LFIAs are qualitative, many prospective quantitative LFIAs have been demonstrated in recent years such as those based on smartphone detection. Another prospective development route has been the development of electrochemical immunoassays.
Faster and more accurate results are highly achievable via the use of current technology. Immunochemistry analyzers have become highly automated over the years. In an effort to reduce human errors and enhance speed, machines with advanced capabilities and high throughput rate are now dominating the industry. Laboratories with investments on these instruments are able to improve their productivity level with confidence. Leading medical instrument manufacturers for the device are offering higher flexibility and comprehensive features with their analyzers that include scalability, the simplicity of operations, streamlined maintenance, and inventory management. They also offer the benefits of enhanced procedure consistency that can help reduce training and labor costs. These value propositions are clearly meant to benefit laboratories and healthcare institutions using the device. It also help support better patient care and improve treatment plans.
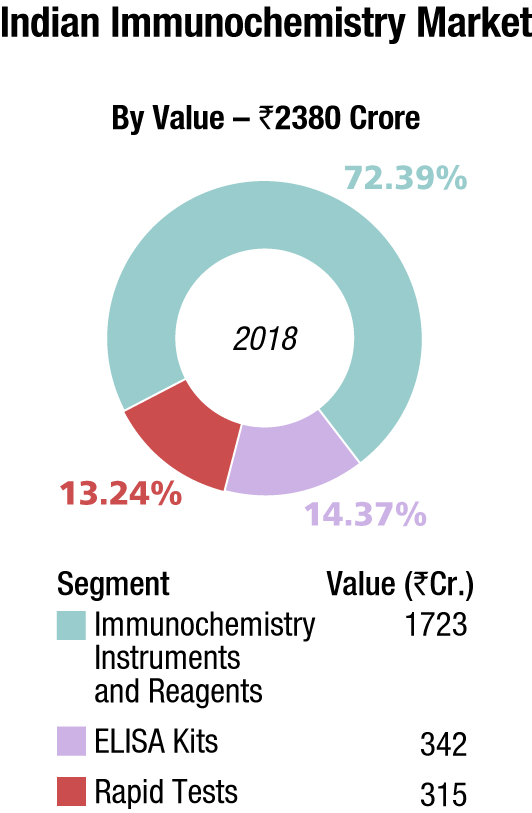
Indian market
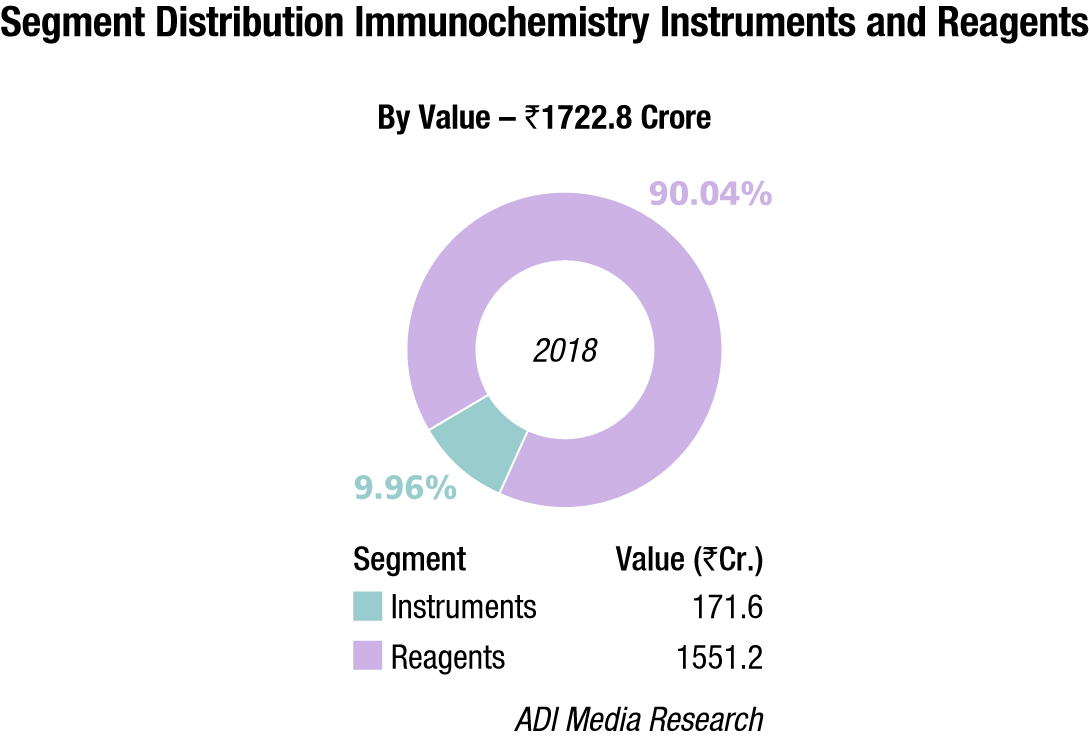
The Indian immunochemistry instruments and reagents market in 2018, is estimated at Rs 1722.8 crore, with reagents dominating at Rs 1551.2 crore. Roche, Abbott, Siemens, bioMérieux, Beckman Coulter and J Mitra are the players with a strong presence. OCD, Tosho, Transasia, and Bio-Rad are aggressive. Other popular brands in this segment are Mindray, Immunoshop, and Maglume. Elisa kits clocked an additional Rs 342 crore and rapid tests, an additional Rs 315 crore in 2018.
As GST implementation in 2017 gained momentum, it had a negative impact on the instruments market, which in turn translated to a slowing in demand in the reagents business in 2018.
Total lab automation has somewhat lost pace over the last couple of years, with a tussle for supremacy among the leading brands. The customers find the business requires too much space and with low ROIs perceive it to be an expensive proposition, not always worthwhile increasing investing in.
Having said that, the emerging trend of corporate players establishing diagnostic centers in small towns and rural areas tends to provide opportunities for the import of automated systems and reagents. For instance, Metropolis Healthcare has announced plans to add 800 collection centers, and 10 labs by end-FY19. Oncquest Laboratories also plans to add 18 more labs across India by 2020-end, mainly in Tier-II cities as the group already has a presence in the Tier-I cities. It is looking at cities such as Vijayawada, Jammu, Jalandhar, Kanpur, Jodhpur, Surat, Bhopal, Indore, Ranchi, Coimbatore, etc., for expansion. Also on the cards is a network of 500 more collection centers taking the total count to 1000 centers.
Vendor update
Mindray has launched one of the world’s fastest chemiluminescenceimmunoassay analyzer, the CL-6000i. The vendor launched it at the China International Medical Equipment Fair (CIMEF Spring 2018). Mindray has also introduced its new laboratory solution, the SAL 9000 Chemi-immuno modular system at the event. Consolidating the BS-2000M chemistry analyzer with its chemiluminescence immunoassay analyzer CL-6000i, the SAL 9000 offers speedy, integrated workstation for clinical chemistry and immunoassay testing with a comprehensive test menu.
| Tier I | Tier II | Tier III | Tier IV | Others |
|---|---|---|---|---|
| Roche | Abbott, Siemens, bioMérieux, Beckman Coulter, and J Mitra | OCD, Tosho, Transasia, and Bio-Rad | Mindray, Maglumi (SNIBE and Immunoshop) | Agappe, CPC, and Meril |
| *Vendors are placed in different tiers on the basis of their sales contribution to the overall revenues of the Indian immunochemistry instruments and reagents market. | ||||
| ADI Media Research | ||||
Sysmex Corporation launched its automated high-sensitive immunoassay system for research applications HI-1000 in March 2018, with the aim of realizing personalized medicine through liquid biopsy. The HI-1000 measures with high degree of sensitivity minute amounts of proteins contained in the blood and other bodily fluids. With this system, the company aims to contribute to the development of new biomarkers for personalized medicine. The HI-1000 is a highly sensitive, fully automated immunoassay system for research use, which combines CLEIA with the immune complex transfer method, achieving a high level of sensitivity by reducing background noise stemming from non-specific binding. This method achieves sensitivity levels ranging from several tens to several thousands of times higher than conventional methods, allowing proteins to be detected at levels of less than one pg/mL. This sensitivity promotes R&D into the measurement of blood biomarkers in minute quantities that were difficult to measure in the past.
Thermo Fisher Scientific received FDA clearance for its de novo submission of the Thermo Scientific QMS Plazomicin immunoassay in December 2018. This quantitative immunoassay measures the concentration of a novel therapeutic antibiotic, Plazomicin. The new QMS Plazomicin immunoassay is the latest addition to the Thermo Fisher line of therapeutic drug monitoring (TDM) immunoassays, which employ proprietary technology across a range of drug classes including those for antibiotics, antiepileptics, antiarrhythmics, immunosuppressants, and others. Thermo Scientific TDM immunoassays enable physicians to monitor and administer therapeutics on an individual patient basis. By maintaining optimized drug levels, the effects of under or overdosage may be minimized and patient outcome improved. This immunoassay was developed in collaboration with Achaogen, Inc. QMS TDM immunoassays are liquid, ready-to-use, and designed to provide precise and accurate measurement of drug levels in patients.
In July 2018, Becton, Dickinson and Company (BD) commercially released the BD AbSeq assay to analyze protein expression at the single-cell level using high-throughput sequencing. The assay brings together high-quality antibodies from the BD Pharmingen portfolio with oligonucleotides, allowing researchers to perform single-cell protein analysis on the BD Rhapsody single-cell analysis system. When used together, these research tools enable researchers to simultaneously analyze RNA and proteins in thousands of individual cells and develop a more complete picture of the role genes and proteins play in biological systems. The product leverages 40 years of BD leadership in immunology research. To advance knowledge of the immune system, BD empowers immunology researchers with a range of tools for multi-omics analysis. The launch of the BD AbSeq assay is the latest innovation in BD’s single cell multi-omics portfolio. Over the last year, BD has built on its BD Rhapsody single-cell analysis system, a complete system of reagents, instruments, software, and targeted gene panels, to offer additional single-cell analysis capabilities.
In November 2018, bioMérieux acquired a majority holding in China-based Suzhou Hybiome Biomedical Engineering Co. Ltd. to strengthen its presence in the immunoassay market. Founded in 2009, Hybiome is specialized in automated immunoassay tests. The company develops, manufactures and markets a complete range of diagnostic solutions (reagents, instruments and software) cleared by the China Food and Drug Administration (CFDA). Hybiome markets two automated, mid-throughput immunoassay systems (AE-180 and AE-240) using the latest generation CLIA technology and featuring a menu of nearly 80 parameters. Hybiome also recently received CFDA clearance for the AE-120 system, to further increase access to its menu of assays. bioMérieux is delighted to enter into this agreement with Hybiome, a company with remarkable expertise in the field of immunoassays. This new important step is fully in line with bioMérieux international development strategy, which consists of locating research and production as close as possible to patients’ and healthcare needs.
Also, in April 2018, bioMérieux acquired Astute Medical Inc. reinforcing its offering of high medical value immunoassay biomarkers. Astute is dedicated to improving the diagnosis of high-risk medical conditions and diseases through the identification and validation of protein biomarkers. In particular, the company developed the Nephrocheck test, an FDA-cleared test for the early risk assessment of acute kidney injuries based on the level of two biomarkers, IGFBP-7 and TIMP-2. This acquisition was built upon the fruitful partnership developed in 2015 between Astute Medical and bioMérieux when Astute granted bioMérieux a license to develop and market the Nephrocheck test for the VIDAS automated immunoassay system. Since 2017, bioMérieux has been a licensed distributor with Astute for the Nephrocheck test on the Astute140 Meter in the US. bioMérieux intends to continue to invest in health economic and outcome studies for Nephrocheck, explore the other promising biomarkers in the Astute pipeline as well as work with Astute’s current license and distribution partners in order to make the Nephrocheck test available to as many patients as possible worldwide.
Global market
Immunochemistry analyzers are becoming more and more popular globally as these are proving to be a very effective tool to diagnose cancer, hepatitis, illegal drugs, fertility problems, sodium levels, endocrine function, and the detection of blood clots. Globally, the immunochemistry market represents approximately 40.1 percent of the global in vitro diagnostics (IVD) market. Immunochemistry is by far the largest segment by volume, mostly due to routine testing. It is primarily driven by growing access to healthcare. It is very important to know which analyzer to use as different analyzers have different analysis time, and need different reagents which make these instruments highly sophisticated. With the rapid technological advancement in healthcare industry, the immunochemistry market is expected to grow from USD 1.69 billion in 2018 to USD 2.77 billion by 2025, reflecting a healthy CAGR of 7.27 percent, according to MarketInsightsReports.
The increasing incidence of chronic and infectious diseases, technological advancements in immunochemistry instruments and introduction of novel automated systems, growth in the biotechnology and biopharmaceutical industries, increasing adoption of immunoassay-based point-of-care testing and rapid testing, and the vital role of immunoassays in drug and alcohol testing are the major factors driving the growth of this market. On the other hand, stringent regulatory requirements for the approval of the instruments and consumables and the implementation of excise duty by the US government are expected to restrain the growth of this market.
Under the strong rules and regulation laboratories find it difficult to have good revenues. Thus, it is very important to find ways to sustain in such a cost-crunched environment. Due to the cost cutting in clinical lab fees, profitability per test is decreasing which makes it necessary for the laboratories to focus on the volume rather than the value. There is also heavy pressure for quality, error-free results to ensure patient satisfaction. This forces the labs to lean toward more automated systems with effective workflow solutions. In US, laboratories are encouraged for automation due to the heavy influx of patients with insurance coverage. But with a limited workforce in the clinical lab, it is difficult to manage the huge inflow of patients. On the other hand, patients need prompt and error-free results. Thus, this scenario demands laboratories to seek the help of systems that have high accuracy, with managing high growing volumes and also offer remote data acquisition capabilities. The automated analyzers are available with barcode readers, rack detection systems, and sample/plate identification modules to avoid plate or sample switch.
Demand for immunochemistry analyzers is slowing in the US and Western and Eastern Europe. Europe’s challenges in this segment include laboratory consolidation in France and economically troubled Greece, Italy, Spain, and Portugal. China’s growing rural hospital market lacks basic diagnostics laboratory infrastructure and represents an untapped opportunity for affordable immunochemistry analyzers.
The market is likely to witness a stiff competition among the prominent players that are engaged in the market across the globe. The high development of the medical industry and the increasing investments by the players for research activities are projected to ensure the development of the market in the next few years. Moreover, the high number of collaborations and acquisitions is considered as another key factor that is predicted to ensure the market development in the coming years. The leading players in the global market include Siemens Healthineers, Abbott Laboratories, Beckman Coulter, Ortho-clinical Diagnostics, Thermo Fisher Scientific, F. Hoffmann-La Roche, Becton, Dickinson and Company, Danaher Corporation, Sysmex Corporation, Mindray, bioMérieux, and DiaSorin.
Technological trends
Immunochemistry is one of the oldest, most well-known, and widely used bioanalytical technologies. Various methods in immunochemistry have been developed and refined and been used in scientific study, from virology to molecular evolution. Significant advances in immunochemistry technologies during the last few decades have drastically shortened the duration of a procedure manifold in comparison with conventional immunoassays. The evolution of microfluidics and LOC technologies based on automated microfluidic protocols has resulted in prospective semi- and fully automated immunoassays, enabling point-of-care testing (PoCT) of analytes at remote settings. As the number of steps in the immunoassays has also been critically reduced, a wide range of rapid immunoassays has resulted in novel formats to detect analytes in less than 30 minutes. The use of emerging micro- and nanomaterials has paved the way for immunoassays to achieve detectivity in the range of fg/mL. Such low or even lower detection limits are needed for early detection of cancer or the prompt detection of relapse after cancer treatment and therapy. To date, advances together with novel detection concepts such as chemiluminescent resonance energy transfer and fluorescence resonance energy transfer have further pushed down the limits of sensitivity to attogram levels. The use of nanomaterials and novel concepts results in the emergence of visualized immunoassays, which can read and analyze pertinent information based on visual detection. This interesting trend in immunodiagnostics has instigated an interest toward the development of instrument-free immunoassays.
There is also an emerging trend toward the development of multiplexed analyzers that holds tremendous potential in IVD and bioanalytical sciences. Other new biosensing technologies are based on microgravimetry (cantilevers and quartz crystal microbalance), interferometry, and acoustic waves. Although such formats have been demonstrated in various laboratories, they could not gather much commercial attention. Of interest is the deployment of mass spectroscopy–based immunoassays, which have been extensively used as the clinical and industrial immunoassay standard for a large number of analytes. Albeit it requires highly sophisticated and costly instrumentation along with highly skilled analysts, this technique offers unmatched high sensitivity, specificity, and analytical performance. Accordingly, it is being used as the established clinically validated assay for various analytes, which makes it essential to use these immunoassays to determine the precision of newly developed analyzers. Other parallel improvements are antibody immobilization chemistries, rapid immunoassay strategies, portable readout devices, innovative diagnostic platforms, and assisting complementary technologies. These ongoing advances have considerably improved the bioanalytical performance of analyzers and would further lead to continuous improvements in immunochemistry analyzers during the coming years.
The surface plasmon resonance (SPR)-based instruments for label-free immunoassays are another striking advance, leading to the emergence of real-time label-free assays. These systems have become the global standard to probe biomolecular interactions, the development of rapid immunoassays for analytes, and screening of appropriate immunological components. The main characteristic features of SPR-based analyzers are critically reduced the sample-to-answer time of only a few minutes; minimal samples and reagents requirement of only tens of microliters; a label-free microfluidic procedure; and a fully automated analysis with a high throughput. Moreover, a large number of surface-functionalized SPR chips, immobilization chemistries, regeneration buffers, and assay protocols further facilitate the rapid development of SPR-based assays. The low-cost formats including the disposable paper-based immunoassays have been the long-cherished vision as they would be ideal for the developing nations and remote settings. The developed complementary technologies during the last decade have led to the emergence of several prospective paper-based assays. However, they still require intensive efforts as their bioanalytical performance is not optimal for real sample testing and inferior compared with that of the established formats.
Conventional immunochemistry technologies are laborious, time-consuming, and expensive. To address these limitations, researchers have been developing miniature biosensors which offer enhanced simplicity, faster measurement times, and lower costs compared with benchtop diagnostic assays. Of all biosensing modalities, electrochemical detection is one of the most popular due to its excellent analytical performance, simplicity, portability and low costs. These features also make electrochemical sensors well suited for point-of-care applications, such as health monitoring (e.g. glucose testing) and IVD testing in resource-limited settings. While these devices are promising, many of them require protein labeling and/or washing, which complicates and lengthens the testing process. Additionally, most immunoassays utilize enzymes for signal transduction or amplification, which results in limited stability at room temperature.
The most recent trend is the development of smartphone (SP)-based immunoassays that have been developed with superior bioanalytical performances and capabilities far exceeding those performed using advanced central laboratory instruments. The use of SPs enhances the capabilities of diagnostics and enables the analysis at the PoC settings, such as decentralized and remote locations, and personalized settings. The SP-based immunoassays can be performed at any place and time without the need for a continuous power supply, highly skilled analysts, and healthcare professionals. The readings together with the time and date stamp and spatiotemporal information can be stored securely on the SP and the centralized server by cloud computing via wireless or Internet connectivity. The readings can be immediately processed on the SP or the centralized server by authorized healthcare professionals and the results are transmitted back to the user. SPs have emerged as the ideal PoC device for immunochemistry as they are extensive outreach and capabilities considering over 7 billion SP users, i.e., 96 percent of the world’s population.
With the help of recent computer technology, biochemical testing has taken a new leap in turnaround time and accuracy. It has opened up new treatment possibilities and provided clearer pictures of patients’ health issues. Newly developed immunochemistry analyzers with computer technology are mission-critical in nature and each requires the use of an industrial computer that is capable of operating 24×7 with high dependability and also with matching processing power to perform complex tasks. Embedded motherboards or systems are integrated into these devices to control their operations. Touch panel PCs or display monitors made specifically for healthcare environments are also integrated for use as a user interface for the analyzers. These motherboards, embedded systems, and touch panel computers offer rich I/Os that are designed for expansion and ease of customization. The considerations also include future-proofed computer solutions with high interoperability with software and other peripherals. These analyzers also offer a variety of value-added features. For example, some analyzers come with refrigerated storage for the reagents and test subjects for added convenience and time-saving which, in turn, result in higher productivity level. Some offer specific features and reporting to work with specialized assays. All features require interoperability among the analyzer’s hardware, software, and other peripherals and are crucial for the analyzer to deliver its complete capabilities.
Road ahead
As medical science has come to understand that the human body is controlled on the molecular level by various proteins, hormones, drugs, and other substances, technologies have developed to detect levels of these molecules in order to monitor health and diagnose disease. However, many of these molecules are so small that they cannot be detected by the most widely available analysis techniques, leaving questions about crucial substances like amino acids, sugars, and lipids largely unanswered. Scientists at the Wyss Institute for Biologically Inspired Engineering at Harvard University together with Brigham and Women’s Hospital (BWH) have created a new type of immunoassay that is capable of detecting small molecules with 50-fold greater sensitivity than conventional detection methods, and can be easily integrated into existing diagnostic platforms. The enhanced analytical sensitivity enables measurements of small molecules at extremely low concentrations, and opens a window into biological phenomena that were previously unreachable. The scientists are planning to use this method in diagnostics for improved detection of hormones in blood samples and are working to try to commercialize this technology for the rapid detection of small molecules for a variety of clinical applications. By sensing molecules previously undetectable within an hour, they will open entirely new approaches to diagnostics and clinical monitoring that should greatly improve human health!
The coming years are anticipated to witness continuous innovation and breakthroughs in immunochemistry platforms and complementary technologies. The next-generation analyzers, based on highly simplified immunoassay formats will require only minimal process steps and critically reduced sample volumes without any washing steps. They will be fully automated, cost-effective, and equipped with a smartphone- or gadget-based mobile healthcare capabilities. Therefore, they would be deployed at centralized, decentralized, remote, and personalized settings with minimal or no requirement for power supply and skilled analysts. The trend is strongly inclined toward immunoassay formats to perform multiplex detection of biomarkers for a particular disease and offer clinical diagnosis and monitoring effectively via clinical score. The evolving trend toward SP-based technologies can extensively extend the outreach of diagnostics by providing personalized mobile healthcare monitoring and management.
Industry Speak
Immunoassay Instruments Market Overview
Dharampal Sharma
Product Manager – Immunoassay
Mindray Medical India Private Limited
The Indian immunoassay instruments market is expected to witness considerable growth in the coming years, mainly due to the rising prevalence of chronic and infectious diseases.
Among immunoassay technologies, chemiluminescence held the largest share in the immunoassay instruments market, due to the wide adoption of this testing technique for detecting chronic and infectious diseases.
Factors driving the immunoassay instruments market include the discovery of specific biomarkers, extensive use of immunoassays in oncology, rising geriatric population, and the growing preference for personalized medicine. Increasing research activities focused on exploring the potential application of immunoassay tests in identifying infectious diseases, toxicology testing, and other drug monitoring tests are some other factors that are expected to drive the market growth in the coming years. Furthermore, the key market players are continuously involved in the development of compact, reliable and portable products, as a result of technological advancements, which is also supporting the market demand.
Mindray a new player in immunoassay market
As a major player in the field of in vitro diagnostics, Mindray is committed to contributing to the improvement of public health and it has enabled to extend Mindray’s expertise to the fields of immunology through innovative chemiluminescence technology with development of varying ranges of immunoassay instrumentation. We have immunoassays solutions for all the laboratory segments from core laboratories (SAL 8000 and 9000) to centralized laboratories (CL-6000i) to medium laboratories (CL-2000i, CL-1200i, and CL- 1000i) to entry-level laboratories (CL-900i and CL-960i). We intend to meet the demand in emerging markets for immunoassays testing by providing a high-value-added testing environment comprising of compact, highly functional, the unique combination of instrument and reagents that deliver high clinical value. Going forward, the company aims to reduce the burden on patients and enhance the quality of testing, as well as instilling its customers with confidence.
Industry Speak
Cartridge-based Nephelometry And Immunology Testing Systems Are Gaining Momentum
John Mathayi
Group Product Manager – Nephelometry and Hematology
Agappe Diagnostics Ltd.
The rise of phoenix is the right connotation for field of immunology, which is experiencing a period of extraordinary advancement in both Immunochemistry Instrumentation segment and reagent segment. Immunochemistry analyzers are becoming more and more popular nowadays as these have proved to be very effective tools to diagnose of specific proteins related to cancer, renal disorders, life style, and nutritional disorders. The global immunochemistry instruments and reagents market is expected to reach USD 24.5 billion by 2019 from USD 17.3 billion in 2014, growing at a CAGR of 7.2 percent from 2014 to 2019. Biomarkers the fine development across globe support detection of diseases in their initial stage. Immunochemistry plays a crucial role in the development of biomarkers thus with the growing demand of biomarkers, the market for immunochemistry instruments and reagents is also expected to flourish. In addition to this, technological advancements due to which automated, and more accurate instruments are present in the market is another major factor propelling the growth of the market. Cartridge based nephelometry analyzers and cartridge based immunology testing systems are gaining its momentum in Indian diagnostic market because of the simple operation, compact hardware, and smaller pack sizes.
Manufacturers are focusing on, operator convenience, accuracy, specificity, speed, robustness, and sensitivity, also there is a shift of customers from standalone high cost floor model systems, toward highly affordable bench top analyzers with very low recurring cost.
In this global race of technological innovation and quality Agappe Diagnostics Ltd, pioneer in nephelometry and immunochemistry manufacturing and a leading diagnostic reagents and equipment manufacturer in India is focusing to its core in immunochemistry, has announced a tie-up with Japanese multinational Toyobo Co Ltd for technology licensing to manufacture early cardiac, cancer, and sepsis detection device. The compact analyzer is designed with smart based card calibration for error free testing, the upcoming products will be offering unique user interface, high sensitivity with precise and accurate result.












